First 3D Images of Human Embryo Implantation Reveal New Details of the Process
Analyzing embryo movements in uteruslike environments could offer clues to improving the success rate of in vitro fertilization
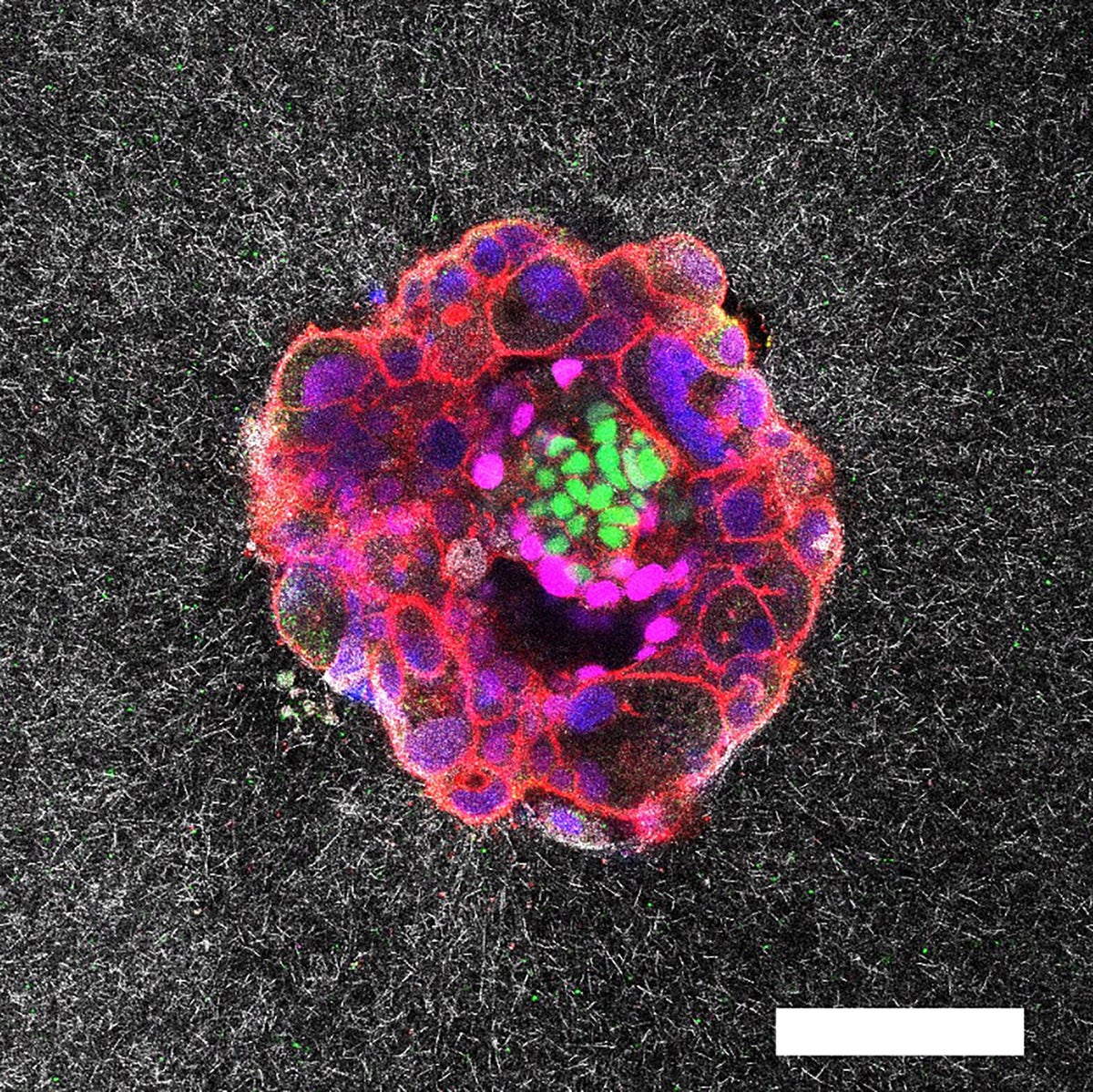
Confocal microscopy image of a nine-day-old human embryo. Specific proteins and cellular structures have been coloured in the image: OCT4 (green), which is related to embryonic stem cells; GATA6 (magenta), which is associated with early tissue formation; DAPI (blue), which marks the DNA in the nuclei; and phalloidin (red), which reveals the actin cytoskeleton. The scale bar corresponds to 100 µm.
Researchers have captured the very first real-time, three-dimensional images and videos of a human embryo implanting into collagen designed to mimic uterine tissue —a key stage in reproduction. The resulting footage, which shows how embryos push and pull to anchor themselves in the uterus in vivid detail, could lead to improvements for in vitro fertilization (IVF) techniques, the scientists say.
Five days after an embryo is fertilized artificially, fertility doctors must implant it into the body so it can continue to grow. “What happens between the transfer and the first ultrasound weeks later is a black box,” says Ojosnegros, who is also co-founder of the biotech company Serabiotics. Implantation failure is one of the main causes of infertility —up to 60 percent of miscarriages occur during this process.
If you’re enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
The first successful culture of human embryos beyond implantation was demonstrated in a petri dish in a lab in 2016, but Ojosnegros and his team wanted to see what this process would look like in 3D tissue that was more similar to that of the uterus.
To do this, the team designed a special ex vivo system made of gel and collagen—a protein found in the uterine lining—and used embryos donated by people who had completed an assisted reproduction process. The system works, Ojosnegros says, because the network of collagen fibers signals to the embryo at a molecular level that this is a natural matrix.
By using advanced 3D microscopes, the researchers recorded the action over time. Tracking tiny movements in the gel’s fibers allowed them to map exactly where and how strongly the embryos were pulling. The researchers did the same with mouse embryos to compare movement patterns.
The footage showed that human embryos generate a network of tiny pulling forces that ripple through the womb. They burrow into the surrounding tissue from one side, creating multiple small traction points that tug the lining in all directions. Mouse embryos, on the other hand, spread out more across the surface and pull mainly along two or three strong lines.
Embryo compacting and invading the uterine tissue.
When the researchers applied external tension to the matrix, tugging it with tiny forceps, they noticed the embryos reoriented toward those areas. The scientists suggest micro contractions might be guiding the embryo to implant in the optimal direction in the uterus. “We believe these micro contractions are what the embryo uses to guide itself toward the blood vessels and the nutrients it needs,” Ojosnegros explains, adding that more studies are needed to confirm this hypothesis.
Two embryos implanting into the uterus.
Source: www.scientificamerican.com